BCVA and refractive diopter 3 months after
surgery were compared with those before surgery. The median pre- and post-operative logMAR BCVA
were significantly different. (Wilcoxon signed rank
test,
=-7.999, P<0.001): 1.4 (range 0.8-2.3) and 0.8 (range 0.1-1.4), respectively. The logMAR
BCVA for group B was lower before surgery (Mann-Whitney test,
=0.043). The
logMAR BCVA for groups A and B were similar after
surgery (Mann-Whitney test,
=0.791). Visual acuity unchanged in 4 eyes of 4 cases (4.49%) and was poorer due to cystoid macular
edema (CME) and uveal effusion in 1 eye of 1 case (1.12%). The degree of hyperopia before surgery
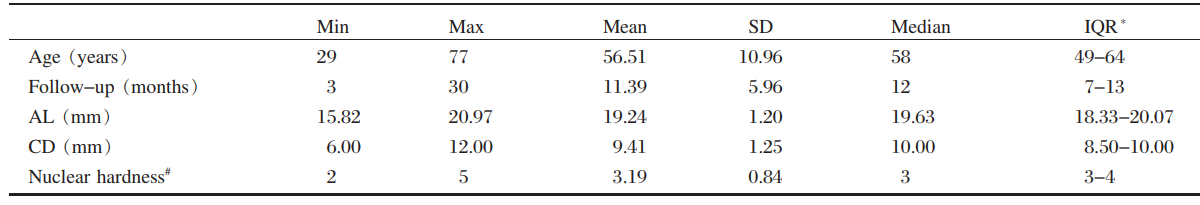
varied from+4.50 D to+20.00 D with a median value
of+9.00 D. After implantation of the IOLs (median
power of 30 D, range 29.00 D to 34.00 D), hyperopia significantly decreased to+3.00 D (range
-0.50 D to 14.00D, Wilcoxon signed rank test,
<0.001). The median difference between
target refractive diopter and achieved postoperative
refractive diopter was +0.50 D (range -1.50 D- +4.00
D), and 37 eyes (41.57%) were within±0.50 D.
Early stage was defined as less and late stage as
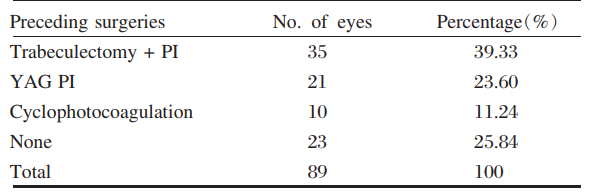
more than 1 month after surgery. Iridoschisis, an intraoperative
complication, was observed in 6 eyes
with glaucoma. Of these, one eye had received
neodymium: YAG laser (YAG) peripheral iridotomy (PI) and 5 eyes had undergone trabeculectomy
and PI. Accordingly, the previous surgeries may
have caused iris trauma and synechia. The most frequent
early postoperative complications were temporary
corneal edema (TCE), AIR, CME and uveal
effusion, while the most frequent late postoperative
complications were CME and uveal effusion. The
incidence of complications were not significantly different
for group A (73.91%) compared to group B (62.12%), (chi-square test, χ2=1.312, P=0.252). Excluding transient TCE and AIR, the incidence of
complications in group A (73.91%) were significantly
higher compared with group B (37.88%, Chisquare
test, χ2=8.887, P=0.003).
The edema and inflammatory response after
surgery were cleared up, and uveal effusion was resolved
in all cases. Treatment of CME was ineffective.
YAG-capsulotomy cleared the posterior capsular
opacification (PCO) in 10 eyes (11.24%) (Table
4). Phacoemulsification surgery did not correct the
elevated IOP in 19 eyes. One eye had normal IOP
and no previous surgical history. Antiglaucomatous
medication was used to manage 18 eyes with high
IOP before surgery, 2 of which developed malignant
glaucoma. Surgical intervention, consisting of YAG
PI, cyclophotocoagulation, trabeculectomy with PI, and YAG-capsulotomy combined with anterior vitrectomy
was performed in 9 eyes (47.37%) which
had high IOP after phacoemulsification surgery. No
other severe complications were detected in the other
10 eyes (52.63%).
The relationship between risk factors and complications
was evaluated. The nuclear hardness of patients
presenting with inflammatory response significantly
differed from that of their counterparts without
inflammatory response (Mann-Whitney test,
P=0.002). PCO was related to AIR (Spearman's rank
correlation,
P=0.000, r=0.42). Univariate and multivariate
logistic regressions were performed to estimate
OR for each variable. Factors were included in
the final multivariate model only if significant (
P<0.05). The data showed nuclear hardness was related
to TCE (
P<0.001, odds ratio [OR]=3.42,95% confidence
interval [CI]=1.75-6.70); AIR was positively
associated with AL (
P=0.003, OR=0.44, 95% CI=0.26-0.76) and nuclear hardness(
P=0.031, OR=0.44, 95% CI=1.08-4.45); AL (P=0.001, OR=0.28, 95%
CI=0.13-0.59), ACA (
P=0.020, OR=0.82, 95% CI=0.70-0.97) Mean keratometry (Km) (
P=0.001, OR=1.82, 95% CI=1.26-2.64) was related to CME. Uveal effusion was related to AL (
P=0.002, OR=
0.01, 95% CI=0.001-0.22).
Table 4 Intraoperative, early and late postoperative complications
TCE=temporary corneal edema; AIR=anterior inflammatory response; CME=cystoid macular edema; PCO=posterior capsular opacification; YAG=neodymium: YAG laser; High IOP=21 mmHg or higher (Goldmann applanation tonometry); PI=peripheral iridotomy.
*Tobramycin+Dexamethasone: topical tobramycin(0.3%)+dexamethasone (0.1%)
#
AIR was documented in the form of flare and/or cells ≥ grade 2 by Hogan's criteria
△Dexamethasone sodium phosphate: intravenous dexamethasone sodium phosphate 5-10 mg daily
Discussion
In our study, there were no complications in the
majority (60.67%) of cases (if TCE and AIR were
excluded), and improvement of BCVA was achieved
in 94.38% of cases. Nonetheless, significant risks are
still associated with phacoemulsification surgery on
nanophthalmic eyes. Compared with a previous study10, we achieved a limited gain in BCVA, with greater
deviation (from 0.1 logMAR to 1.4 logMAR). This
suboptimal visual acuity could be caused by two
factors. Firstly, the majority of patients with nanophthalmos
came from remote rural regions in China, and had poor visual acuity when they were teenagers
or suffered from preexisting ocular diseases such as
refractive amblyopia and foveal underdevelopment. Secondly, most of the eyes included in our study
had glaucoma which could damage the optic nerve
before cataract surgery.
Postoperative BCVA in nanophthalmos was less
favorable compared with that in normal eyes. This
was also true for the refractive results. The target refraction of ±0.50 D was not reached in 52 eyes (58.43%), most of which remained hyperopic. Several
factors may be responsible for this discrepancy, including discrepancies in AL measurement and IOL
power calculation. IOL calculations could be inaccurate, with
the Holladay Ⅱ formula typically being
preferred although it is not readily available11-12. The
Hoffer Q formula which performed as well as the
HolladayⅡ formula for small eyes13 is readily available
and therefore was our method of choice. Although
Gayton et al14 demonstrated 2 IOLs , in a
piggyback manner, can be successfully implanted, we considered that the zonules in those small eyes
may be too weak to support insertion of 2 IOLs. Oshika
et al15 modified the technique by implanting 1 IOL
in the capsular bag and another IOL in the sulcus. We determined that insufficient space was available
for 2 IOLs, and more importantly, that piggyback
IOLs would produce marked hyperopic drift16.
Based on all of these factors, we used a single highpower
IOL instead of piggyback IOLs.
Phacoemulsification lens surgery of nanophthalmic
eyes is associated with a high risk of perioperative
complications, such as corneal decompensation, severe
iritis, explosive choroidal hemorrhage, uveal
effusion, retinal detachment, malignant glaucoma and
CME17. In our cases, TCE, AIR, CME and uveal effusion
accounted for the majority of early postoperative
complications, while CME, uveal effusion glaucoma
and PCO accounted for the majority of late
postoperative complications. Nuclear hardness was a
major risk factor for TCE and AIR. Once CME occurred,
it was difficult to treat.In our study 2 eyes
of 2 patients developed malignant glaucoma, presumably
because of capsular block and aqueous misdirection
and required YAG-capsulotomy combined
with anterior vitrectomy to manage the IOP. Both
patients had intractable IOP elevation (1 eye had cyclophotocoagulation) before
surgery. We deemed that
additional surgical interventions were often required
as soon as complications were detected, emphasizing
the necessity for careful follow-up.
In our study, there were no severe complications. One of the reasons for this may be that the percentage
of high-risk eyes (approximately <18 mm18) was
low (17.98%). More importantly, we believed the
reduction of severe complications was closely related
to surgical technique, and the surgery should always
be handled by an experienced surgeon. To protect the
corneal endothelium, we injected low density vis
coelastic (VISCOAT, Alcon Laboratories Inc, Texas, USA) before capsulorrhexis. The coaxial micro-incision
technique with a 2.2 mm corneal incision reduced
the trauma around the corneal incision site. A
stop and chop technique was performed to divide the
lens as small as possible (approximately 6-8 pieces). We created paracentesis to make a “stab” incision
for the first step, and made the viscoelastic advancing
towards the paracentesis to gradually replace the
aqueous humor. This may help reduce the aqueous
outflow and naturally maintain ACD. We surmised
that a 95 cm bottle height may be suitable, as it
could maintain normal ACD without causing ocular
hypertension or ischemia during surgery. We preferred
topical anesthesia to retrobulbar anesthesia, because it reduced the vitreous pressure. Preoperative
administration of mannitol may be important in preventing
unexpected complications19. As expected, we
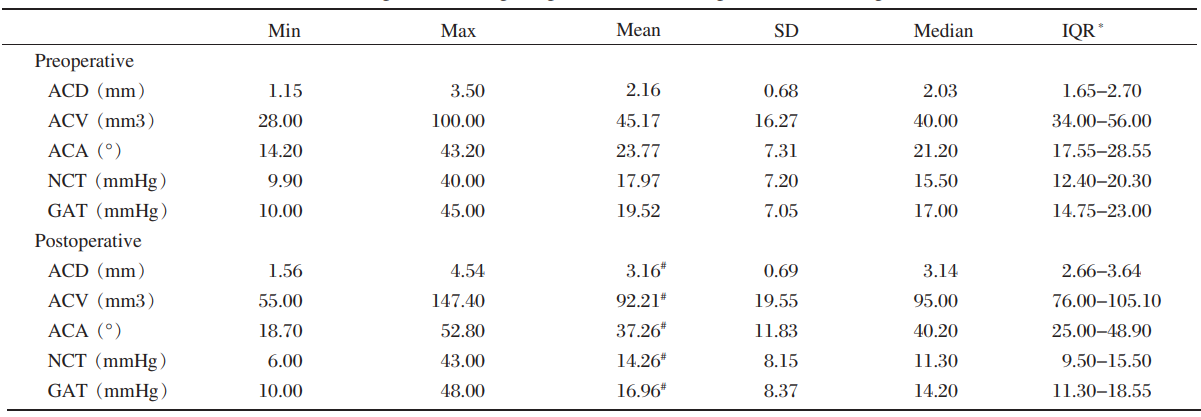
noticed that previous surgical intervention may be
helpful for postoperative ACD and IOP, and may
protect against the development of complications. The risk for complications in nanophthalmos was
greater with reducing AL, ACD, ACV or ACA and
with higher nuclear hardness or Km.
Our study represented a fairly comprehensive case
series. A previous study indicated that when the AL
cut off to define nanophthalmos was <20.9 mm, 0.1
mm less than the 21 mm AL we used to defined
nanophthalmos, the sample size of the present study
would reduce the by nearly 25%20. By and large, the
outcomes that we report were gratifying. Our experience
may be useful to surgeons and patients at the
time of perioperative preparation and preoperative
counseling. The present study was related to the
techniques and skills of an individual surgeon, potentially
limiting the generalization of our results. IOLs
with a power greater than 34 D were not readily
available. This could leave nanophthalmos eyes with
a higher postoperative refractive diopter receiving
less powerful IOLs. Further studies may be more
valuable if we included even short eyes. The decision
to use ocular hypotensive agents was based on clinical
judgment, and patients were not randomized into
groups. We only considered the risk factors correlated
with the surgery when we studied the incidence
of PCO, ignoring the influence of previous holopathy
such as diabetes and hypertension. Therefore, our results may be somewhat biased. Future largescale
multicenter randomized controlled clinical trials
are recommended to generalize our outcomes.
Conclusion
With reasonable preoperative management, prudent
selections of the lens, rigorous surgical technique, and unerring cognition of the potential complications, coaxial
micro-incision phacoemulsification
surgery can be favorably performed in nanophthalmos
with good results. Nonetheless, the surgery
in nanophthalmos is still challenging, primarily due
to the high incidence of complications.