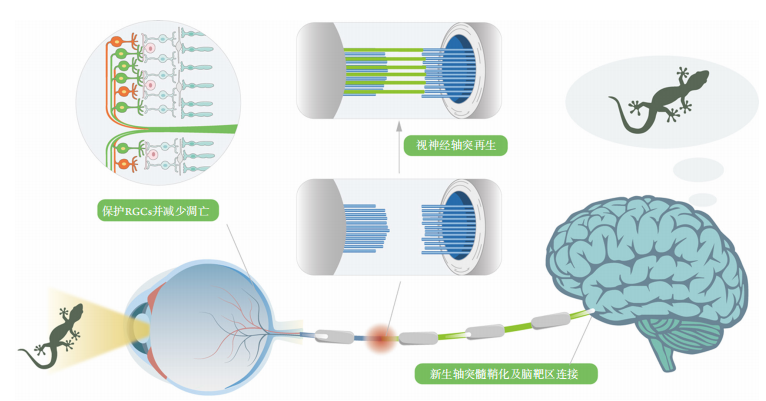
1、Williams PR, Benowitz LI, Goldberg JL, et al. Axon regeneration in the mammalian optic nerve[ J]. Annu Rev Vis Sci, 2020, 6: 195-213.Williams PR, Benowitz LI, Goldberg JL, et al. Axon regeneration in the mammalian optic nerve[ J]. Annu Rev Vis Sci, 2020, 6: 195-213.
2、Weinreb RN, Aung T, Medeiros FA, et al. The pathophysiology and treatment of glaucoma: a review[ J]. JAMA, 2014, 311(18): 1901-1911.Weinreb RN, Aung T, Medeiros FA, et al. The pathophysiology and treatment of glaucoma: a review[ J]. JAMA, 2014, 311(18): 1901-1911.
3、Stein JD, Khawaja AP, Weizer JS, et al. Glaucoma in adults-screening,diagnosis, and management: a review[ J]. JAMA, 2021, 325(2): 164-174.Stein JD, Khawaja AP, Weizer JS, et al. Glaucoma in adults-screening,diagnosis, and management: a review[ J]. JAMA, 2021, 325(2): 164-174.
4、Chauhan MZ, Valencia AK, Piqueras MC, et al. Optic nerve lipidomics reveal impaired glucosylsphingosine lipids pathway in glaucoma[ J]. Invest Ophthalmol Vis Sci, 2019, 60(5): 1789-1798.Chauhan MZ, Valencia AK, Piqueras MC, et al. Optic nerve lipidomics reveal impaired glucosylsphingosine lipids pathway in glaucoma[ J]. Invest Ophthalmol Vis Sci, 2019, 60(5): 1789-1798.
5、Richardson PM, McGuinness UM, Aguayo AJ. Axons from CNS neurons regenerate into PNS grafts[J]. Nature, 1980,284(5753): 264-265.Richardson PM, McGuinness UM, Aguayo AJ. Axons from CNS neurons regenerate into PNS grafts[J]. Nature, 1980,284(5753): 264-265.
6、Benowitz LI, He Z, Goldberg JL, et al. Reaching the brain: advances in optic nerve regeneration[ J]. Exp Neurol, 2017, 287(Pt 3): 365-373.Benowitz LI, He Z, Goldberg JL, et al. Reaching the brain: advances in optic nerve regeneration[ J]. Exp Neurol, 2017, 287(Pt 3): 365-373.
7、Jin Y. Unraveling the mechanisms of synapse formation and axon regeneration: the awesome power of C. elegans genetics[ J]. Sci China Life Sci, 2015, 58(11): 1084-1088.Jin Y. Unraveling the mechanisms of synapse formation and axon regeneration: the awesome power of C. elegans genetics[ J]. Sci China Life Sci, 2015, 58(11): 1084-1088.
8、Huang H, Kaur S, Hu Y, et al. Lab review: molecular dissection of the signal transduction pathways associated with PTEN deletion-induced optic nerve regeneration[J]. Restor Neurol Neurosci, 2019, 37(6): 545-552.Huang H, Kaur S, Hu Y, et al. Lab review: molecular dissection of the signal transduction pathways associated with PTEN deletion-induced optic nerve regeneration[J]. Restor Neurol Neurosci, 2019, 37(6): 545-552.
9、Mak HK, Ng SH, Ren T, et al. Impact of PTEN/SOCS3 deletion on amelioration of dendritic shrinkage of retinal ganglion cells after optic nerve injury[ J]. Exp Eye Res, 2020, 192: 107938.Mak HK, Ng SH, Ren T, et al. Impact of PTEN/SOCS3 deletion on amelioration of dendritic shrinkage of retinal ganglion cells after optic nerve injury[ J]. Exp Eye Res, 2020, 192: 107938.
10、Pita-Thomas W, Mahar M, Joshi A, et al. HDAC5 promotes optic nerve regeneration by activating the mTOR pathway[ J]. Exp Neurol, 2019,317: 271-283.Pita-Thomas W, Mahar M, Joshi A, et al. HDAC5 promotes optic nerve regeneration by activating the mTOR pathway[ J]. Exp Neurol, 2019,317: 271-283.
11、Shum JW, Liu K, So KF, et al. The progress in optic nerve regeneration,where are we?[ J]. Neural Regen Res, 2016, 11(1): 32-36.Shum JW, Liu K, So KF, et al. The progress in optic nerve regeneration,where are we?[ J]. Neural Regen Res, 2016, 11(1): 32-36.
12、Vajda F, Jordi N, Dalkara D, et al. Cell type-specific Nogo-A gene ablation promotes axonal regeneration in the injured adult optic nerve[ J]. Cell Death Differ, 2015, 22(2): 323-335.Vajda F, Jordi N, Dalkara D, et al. Cell type-specific Nogo-A gene ablation promotes axonal regeneration in the injured adult optic nerve[ J]. Cell Death Differ, 2015, 22(2): 323-335.
13、Uesugi N, Kimura Y, Yamashita T, et al. Suppression of the p75 receptor signal attenuates the effect of ephrin-B3 and promotes axonal regeneration of the injured optic nerve[ J]. Cell Death Dis, 2013, 4: e557.Uesugi N, Kimura Y, Yamashita T, et al. Suppression of the p75 receptor signal attenuates the effect of ephrin-B3 and promotes axonal regeneration of the injured optic nerve[ J]. Cell Death Dis, 2013, 4: e557.
14、Benson MD, Romero MI, Lush ME, et al. Ephrin-B3 is a myelin-based inhibitor of neurite outgrowth[ J]. Proc Natl Acad Sci U S A, 2005, 102(30): 10694-10699.Benson MD, Romero MI, Lush ME, et al. Ephrin-B3 is a myelin-based inhibitor of neurite outgrowth[ J]. Proc Natl Acad Sci U S A, 2005, 102(30): 10694-10699.
15、Wang KC, Kim JA, Sivasankaran R, et al. P75 interacts with the Nogo receptor as a co-receptor for Nogo, MAG and OMgp[ J]. Nature, 2002,420(6911): 74-78.Wang KC, Kim JA, Sivasankaran R, et al. P75 interacts with the Nogo receptor as a co-receptor for Nogo, MAG and OMgp[ J]. Nature, 2002,420(6911): 74-78.
16、Mukherjee N, Ghosh S, et al. Myelin associated inhibitory proteins as a therapeutic target for healing of CNS injury[ J]. ACS Chem Neurosci,2020, 11(12): 1699-1700.Mukherjee N, Ghosh S, et al. Myelin associated inhibitory proteins as a therapeutic target for healing of CNS injury[ J]. ACS Chem Neurosci,2020, 11(12): 1699-1700.
17、Goulart CO, Mendon?a HR, Oliveira JT, et al. Repulsive environment attenuation during adult mouse optic nerve regeneration[ J]. Neural Plast, 2018, 2018: 5851914.Goulart CO, Mendon?a HR, Oliveira JT, et al. Repulsive environment attenuation during adult mouse optic nerve regeneration[ J]. Neural Plast, 2018, 2018: 5851914.
18、Pearson CS, Mencio CP, Barber AC, et al. Identification of a critical sulfation in chondroitin that inhibits axonal regeneration[ J]. Elife,2018, 7: 37139.Pearson CS, Mencio CP, Barber AC, et al. Identification of a critical sulfation in chondroitin that inhibits axonal regeneration[ J]. Elife,2018, 7: 37139.
19、Kim J, Sajid MS, Trakhtenberg EF, et al. The extent of extra-axonal tissue damage determines the levels of CSPG upregulation and the success of experimental axon regeneration in the CNS[ J]. Sci Rep,2018, 8(1): 9839.Kim J, Sajid MS, Trakhtenberg EF, et al. The extent of extra-axonal tissue damage determines the levels of CSPG upregulation and the success of experimental axon regeneration in the CNS[ J]. Sci Rep,2018, 8(1): 9839.
20、Lin JZ, Duan MR, Lin N, et al. The emerging role of the chondroitin sulfate proteoglycan family in neurodegenerative diseases[ J/OL]. Rev Neurosci, 2021, Epub ahead of print.Lin JZ, Duan MR, Lin N, et al. The emerging role of the chondroitin sulfate proteoglycan family in neurodegenerative diseases[ J/OL]. Rev Neurosci, 2021, Epub ahead of print.
21、Zhang J, Liu W, Zhang X, et al. Sema3A inhibits axonal regeneration of retinal ganglion cells via ROCK2[ J]. Brain Res, 2020, 1727: 146555.Zhang J, Liu W, Zhang X, et al. Sema3A inhibits axonal regeneration of retinal ganglion cells via ROCK2[ J]. Brain Res, 2020, 1727: 146555.
22、Gottschling C, Wegrzyn D, Denecke B, et al. Elimination of the four extracellular matrix molecules tenascin-C, tenascin-R, brevican and neurocan alters the ratio of excitatory and inhibitory synapses[ J]. Sci Rep, 2019, 9(1): 13939.Gottschling C, Wegrzyn D, Denecke B, et al. Elimination of the four extracellular matrix molecules tenascin-C, tenascin-R, brevican and neurocan alters the ratio of excitatory and inhibitory synapses[ J]. Sci Rep, 2019, 9(1): 13939.
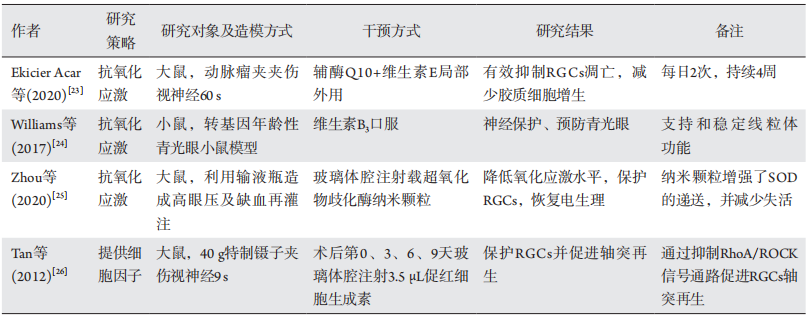
23、Ekicier Acar S, Sar?cao?lu MS, ?olak A, et al. Neuroprotective effects of topical coenzyme Q10 + vitamin E in mechanic optic nerve injury model[ J]. Eur J Ophthalmol, 2020, 30(4): 714-722.Ekicier Acar S, Sar?cao?lu MS, ?olak A, et al. Neuroprotective effects of topical coenzyme Q10 + vitamin E in mechanic optic nerve injury model[ J]. Eur J Ophthalmol, 2020, 30(4): 714-722.
24、Williams PA, Harder JM, Foxworth NE, et al. Vitamin B3 modulates mitochondrial vulnerability and prevents glaucoma in aged mice[ J].Science, 2017, 355(6326): 756-760.Williams PA, Harder JM, Foxworth NE, et al. Vitamin B3 modulates mitochondrial vulnerability and prevents glaucoma in aged mice[ J].Science, 2017, 355(6326): 756-760.
25、Zhou X, Lv J, Li G, et al. Rescue the retina after the ischemic injury by polymer-mediated intracellular superoxide dismutase delivery[ J].Biomaterials, 2021, 268: 120600.Zhou X, Lv J, Li G, et al. Rescue the retina after the ischemic injury by polymer-mediated intracellular superoxide dismutase delivery[ J].Biomaterials, 2021, 268: 120600.
26、Tan H, Zhong Y, Shen X, et al. Erythropoietin promotes axonal regeneration after optic nerve crush in vivo by inhibition of RhoA/ROCK signaling pathway[J]. Neuropharmacology, 2012, 63(6): 1182-1190.Tan H, Zhong Y, Shen X, et al. Erythropoietin promotes axonal regeneration after optic nerve crush in vivo by inhibition of RhoA/ROCK signaling pathway[J]. Neuropharmacology, 2012, 63(6): 1182-1190.
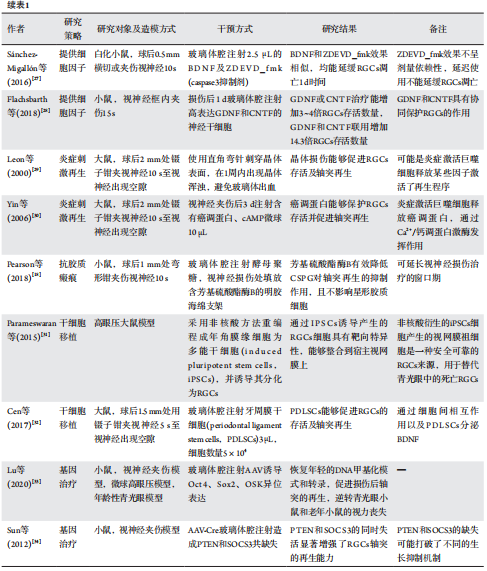
27、Sánchez-Migallón MC, Valiente-Soriano FJ, Nadal-Nicolás FM, et al. Apoptotic retinal ganglion cell death after optic nerve transection or crush in mice: delayed RGC loss with BDNF or a caspase 3 inhibitor[ J]. Invest Ophthalmol Vis Sci, 2016, 57(1): 81-93.Sánchez-Migallón MC, Valiente-Soriano FJ, Nadal-Nicolás FM, et al. Apoptotic retinal ganglion cell death after optic nerve transection or crush in mice: delayed RGC loss with BDNF or a caspase 3 inhibitor[ J]. Invest Ophthalmol Vis Sci, 2016, 57(1): 81-93.
28、Flachsbarth K, Jankowiak W, Kruszewski K, et al. Pronounced synergistic neuroprotective effect of GDNF and CNTF on axotomized retinal ganglion cells in the adult mouse[ J]. Exp Eye Res, 2018, 176: 258-265.Flachsbarth K, Jankowiak W, Kruszewski K, et al. Pronounced synergistic neuroprotective effect of GDNF and CNTF on axotomized retinal ganglion cells in the adult mouse[ J]. Exp Eye Res, 2018, 176: 258-265.
29、Leon S, Yin Y, Nguyen J, et al. Lens injury stimulates axon regeneration in the mature rat optic nerve[ J]. J Neurosci, 2000, 20(12): 4615-4626.Leon S, Yin Y, Nguyen J, et al. Lens injury stimulates axon regeneration in the mature rat optic nerve[ J]. J Neurosci, 2000, 20(12): 4615-4626.
30、Yin Y, Henzl MT, Lorber B, et al. Oncomodulin is a macrophage-derived signal for axon regeneration in retinal ganglion cells[ J]. Nat Neurosci, 2006, 9(6): 843-852.Yin Y, Henzl MT, Lorber B, et al. Oncomodulin is a macrophage-derived signal for axon regeneration in retinal ganglion cells[ J]. Nat Neurosci, 2006, 9(6): 843-852.
31、Parameswaran S, Dravid SM, Teotia P, et al. Continuous non-cell autonomous reprogramming to generate retinal ganglion cells for glaucomatous neuropathy[ J]. Stem Cells, 2015, 33(6): 1743-1758.Parameswaran S, Dravid SM, Teotia P, et al. Continuous non-cell autonomous reprogramming to generate retinal ganglion cells for glaucomatous neuropathy[ J]. Stem Cells, 2015, 33(6): 1743-1758.
32、Cen LP, Ng TK, Liang JJ, et al. Human periodontal ligament-derived stem cells promote retinal ganglion cell survival and axon regeneration after optic nerve injury[ J]. Stem Cells, 2018, 36(6): 844-855.Cen LP, Ng TK, Liang JJ, et al. Human periodontal ligament-derived stem cells promote retinal ganglion cell survival and axon regeneration after optic nerve injury[ J]. Stem Cells, 2018, 36(6): 844-855.
33、Lu Y, Brommer B, Tian X, et al. Reprogramming to recover youthful epigenetic information and restore vision[ J]. Nature, 2020, 588(7836):124-129.Lu Y, Brommer B, Tian X, et al. Reprogramming to recover youthful epigenetic information and restore vision[ J]. Nature, 2020, 588(7836):124-129.
34、Sun F, Park KK, Belin S, et al. Sustained axon regeneration induced by co-deletion of PTEN and SOCS3[J]. Nature, 2011,480(7377): 372-375.Sun F, Park KK, Belin S, et al. Sustained axon regeneration induced by co-deletion of PTEN and SOCS3[J]. Nature, 2011,480(7377): 372-375.
35、Zanon-Moreno V, Ortega-Azorin C, Asensio-Marquez EM, et al.A multi-locus genetic risk score for primary open-angle glaucoma(POAG) variants is associated with POAG risk in a mediterranean population: inverse correlations with plasma vitamin C and E concentrations[ J]. Int J Mol Sci, 2017, 18(11): 2302.Zanon-Moreno V, Ortega-Azorin C, Asensio-Marquez EM, et al.A multi-locus genetic risk score for primary open-angle glaucoma(POAG) variants is associated with POAG risk in a mediterranean population: inverse correlations with plasma vitamin C and E concentrations[ J]. Int J Mol Sci, 2017, 18(11): 2302.
36、Wan P, Su W, Zhang Y, et al. Trimetazidine protects retinal ganglion cells from acute glaucoma via the Nrf2/Ho-1 pathway[ J]. Clin Sci(Lond), 2017, 131(18): 2363-2375.Wan P, Su W, Zhang Y, et al. Trimetazidine protects retinal ganglion cells from acute glaucoma via the Nrf2/Ho-1 pathway[ J]. Clin Sci(Lond), 2017, 131(18): 2363-2375.
37、Chen YJ, Huang YS, Chen JT, et al. Protective effects of glucosamine on oxidative-stress and ischemia/reperfusion-induced retinal injury[ J].Invest Ophthalmol Vis Sci, 2015, 56(3): 1506-1516.Chen YJ, Huang YS, Chen JT, et al. Protective effects of glucosamine on oxidative-stress and ischemia/reperfusion-induced retinal injury[ J].Invest Ophthalmol Vis Sci, 2015, 56(3): 1506-1516.
38、?en M, Akbay?r E, Mercan ?, et al. Cytokine-chemokine and cognitive profile of multiple sclerosis patients with predominant optic nerve and spinal cord involvement[ J]. J Spinal Cord Med, 2021, 44(3): 411-417.?en M, Akbay?r E, Mercan ?, et al. Cytokine-chemokine and cognitive profile of multiple sclerosis patients with predominant optic nerve and spinal cord involvement[ J]. J Spinal Cord Med, 2021, 44(3): 411-417.
39、Kashkouli MB, Yousefi S, Nojomi M, et al. Traumatic optic neuropathy
treatment trial (TONTT): open label, phase 3, multicenter, semi-
experimental trial[ J]. Graefes Arch Clin Exp Ophthalmol, 2018,
256(1): 209-218.Kashkouli MB, Yousefi S, Nojomi M, et al. Traumatic optic neuropathy
treatment trial (TONTT): open label, phase 3, multicenter, semi-
experimental trial[ J]. Graefes Arch Clin Exp Ophthalmol, 2018,
256(1): 209-218.
40、Zamani N, Hassanian-Moghaddam H, Shojaei M, et al. Evaluation of the effect of erythropoietin + corticosteroid versus corticosteroid alone in methanol-induced optic nerve neuropathy[ J]. Cutan Ocul Toxicol,2018, 37(2): 186-190.Zamani N, Hassanian-Moghaddam H, Shojaei M, et al. Evaluation of the effect of erythropoietin + corticosteroid versus corticosteroid alone in methanol-induced optic nerve neuropathy[ J]. Cutan Ocul Toxicol,2018, 37(2): 186-190.
41、Kucuk B, Cevik Y, Acar U, et al. Therapeutic potential of erythropoietin in retinal and optic nerve diseases[ J]. CNS Neurol Disord Drug Targets, 2015, 14(9): 1225-1234.Kucuk B, Cevik Y, Acar U, et al. Therapeutic potential of erythropoietin in retinal and optic nerve diseases[ J]. CNS Neurol Disord Drug Targets, 2015, 14(9): 1225-1234.
42、Hernández CC, Burgos CF, Gajardo AH, et al. Neuroprotective effects of erythropoietin on neurodegenerative and ischemic brain diseases: the role of erythropoietin receptor[ J]. Neural Regen Res, 2017, 12(9):1381-1389.Hernández CC, Burgos CF, Gajardo AH, et al. Neuroprotective effects of erythropoietin on neurodegenerative and ischemic brain diseases: the role of erythropoietin receptor[ J]. Neural Regen Res, 2017, 12(9):1381-1389.
43、Shi Q, Gao W, Han X, et al. Collagen scaffolds modified with collagen-binding bFGF promotes the neural regeneration in a rat hemisected spinal cord injury model[ J]. Sci China Life Sci, 2014, 57(2): 232-240.Shi Q, Gao W, Han X, et al. Collagen scaffolds modified with collagen-binding bFGF promotes the neural regeneration in a rat hemisected spinal cord injury model[ J]. Sci China Life Sci, 2014, 57(2): 232-240.
44、Feng L, Puyang Z, Chen H, et al. Overexpression of brain-derived neurotrophic factor protects large retinal ganglion cells after optic nerve crush in mice[ J]. eNeuro, 2017, 4(1): ENEURO.0331-16.2016.Feng L, Puyang Z, Chen H, et al. Overexpression of brain-derived neurotrophic factor protects large retinal ganglion cells after optic nerve crush in mice[ J]. eNeuro, 2017, 4(1): ENEURO.0331-16.2016.
45、Lv XM, Liu Y, Wu F, et al. Human umbilical cord blood-derived stem cells and brain-derived neurotrophic factor protect injured optic nerve:viscoelasticity characterization[ J]. Neural Regen Res, 2016, 11(4):652-656.Lv XM, Liu Y, Wu F, et al. Human umbilical cord blood-derived stem cells and brain-derived neurotrophic factor protect injured optic nerve:viscoelasticity characterization[ J]. Neural Regen Res, 2016, 11(4):652-656.
46、Mohd Lazaldin MA, Iezhitsa I, Agarwal R, et al. Neuroprotective effects of brain-derived neurotrophic factor against amyloid beta 1-40-induced retinal and optic nerve damage[ J]. Eur J Neurosci, 2020, 51(12):2394-2411.Mohd Lazaldin MA, Iezhitsa I, Agarwal R, et al. Neuroprotective effects of brain-derived neurotrophic factor against amyloid beta 1-40-induced retinal and optic nerve damage[ J]. Eur J Neurosci, 2020, 51(12):2394-2411.
47、Duprey-Díaz MV, Blagburn JM, Blanco RE, et al. Changes in fibroblast growth factor-2 and FGF receptors in the frog visual system during optic nerve regeneration[ J]. J Chem Neuroanat, 2012, 46(1/2): 35-44.Duprey-Díaz MV, Blagburn JM, Blanco RE, et al. Changes in fibroblast growth factor-2 and FGF receptors in the frog visual system during optic nerve regeneration[ J]. J Chem Neuroanat, 2012, 46(1/2): 35-44.
48、Unoki K, LaVail MM, et al. Protection of the rat retina from ischemic injury by brain-derived neurotrophic factor, ciliary neurotrophic factor,and basic fibroblast growth factor[ J]. Invest Ophthalmol Vis Sci, 1994,35(3): 907-915.Unoki K, LaVail MM, et al. Protection of the rat retina from ischemic injury by brain-derived neurotrophic factor, ciliary neurotrophic factor,and basic fibroblast growth factor[ J]. Invest Ophthalmol Vis Sci, 1994,35(3): 907-915.
49、Vega-Meléndez GS, Blagburn JM, Blanco RE, et al. Ciliary neurotrophic factor and fibroblast growth factor increase the speed and number of regenerating axons after optic nerve injury in adult Rana pipiens[ J]. J Neurosci Res, 2014, 92(1): 13-23.Vega-Meléndez GS, Blagburn JM, Blanco RE, et al. Ciliary neurotrophic factor and fibroblast growth factor increase the speed and number of regenerating axons after optic nerve injury in adult Rana pipiens[ J]. J Neurosci Res, 2014, 92(1): 13-23.
50、Wang KC, Koprivica V, Kim JA, et al. Oligodendrocyte-myelin glycoprotein is a Nogo receptor ligand that inhibits neurite outgrowth[ J]. Nature, 2002, 417(6892): 941-944.Wang KC, Koprivica V, Kim JA, et al. Oligodendrocyte-myelin glycoprotein is a Nogo receptor ligand that inhibits neurite outgrowth[ J]. Nature, 2002, 417(6892): 941-944.
51、Mesentier-Louro LA, Rosso P, Carito V, et al. Nerve Growth Factor Role on Retinal Ganglion Cell Survival and Axon Regrowth: Effects of Ocular Administration in Experimental Model of Optic Nerve Injury[ J]. Mol Neurobiol, 2019, 56(2): 1056-1069.Mesentier-Louro LA, Rosso P, Carito V, et al. Nerve Growth Factor Role on Retinal Ganglion Cell Survival and Axon Regrowth: Effects of Ocular Administration in Experimental Model of Optic Nerve Injury[ J]. Mol Neurobiol, 2019, 56(2): 1056-1069.
52、李勇. 鼠神经生长因子联合甲泼尼龙治疗外伤性马尾神经损伤临床疗效及安全性研究[ J]. 中国临床药理学杂志, 2016, 32(15):
1381-1383.
LI Yong. Clinical trial of efficacy and safety of mouse nerve growth factor combined with methylprednisolone in the treatment of traumatic injury of cauda equine[ J]. The Chinese Journal of Clinical Pharmacology, 2016, 32(15): 1381-1383.李勇. 鼠神经生长因子联合甲泼尼龙治疗外伤性马尾神经损伤临床疗效及安全性研究[ J]. 中国临床药理学杂志, 2016, 32(15):
1381-1383.
LI Yong. Clinical trial of efficacy and safety of mouse nerve growth factor combined with methylprednisolone in the treatment of traumatic injury of cauda equine[ J]. The Chinese Journal of Clinical Pharmacology, 2016, 32(15): 1381-1383.
53、Zhong Y, Shen X, Liu X, et al. The early effect of nerve growth factor in the management of serious optic nerve contusion[ J]. Clin Exp Optom,2010, 93(6): 466-470.Zhong Y, Shen X, Liu X, et al. The early effect of nerve growth factor in the management of serious optic nerve contusion[ J]. Clin Exp Optom,2010, 93(6): 466-470.
54、Rovere G, Nadal-Nicolás FM, Sobrado-Calvo P, et al. Topical treatment with bromfenac reduces retinal gliosis and inflammation after optic nerve crush[ J]. Invest Ophthalmol Vis Sci, 2016, 57(14): 6098-6106.Rovere G, Nadal-Nicolás FM, Sobrado-Calvo P, et al. Topical treatment with bromfenac reduces retinal gliosis and inflammation after optic nerve crush[ J]. Invest Ophthalmol Vis Sci, 2016, 57(14): 6098-6106.
55、Leibinger M, Andreadaki A, Diekmann H, et al. Neuronal STAT3 activation is essential for CNTF- and inflammatory stimulation-induced CNS axon regeneration[ J]. Cell Death Dis, 2013, 4: e805.Leibinger M, Andreadaki A, Diekmann H, et al. Neuronal STAT3 activation is essential for CNTF- and inflammatory stimulation-induced CNS axon regeneration[ J]. Cell Death Dis, 2013, 4: e805.
56、Yin Y, Cui Q, Gilbert HY, et al. Oncomodulin links inflammation to optic nerve regeneration[ J]. Proc Natl Acad Sci U S A, 2009, 106(46):19587-19592.Yin Y, Cui Q, Gilbert HY, et al. Oncomodulin links inflammation to optic nerve regeneration[ J]. Proc Natl Acad Sci U S A, 2009, 106(46):19587-19592.
57、王秋红, 魏锐利. 外伤性视神经损伤发病机制及治疗进展[ J]. 中华神经外科疾病研究杂志, 2016, 15(2): 190-192.
WANG Qiuhong, WEI Ruili. Progress in pathogenesis and treatment of traumatic optic nerve injury[ J]. Chinese Journal of Neurosurgical Disease Research, 2016, 15(2): 190-192.王秋红, 魏锐利. 外伤性视神经损伤发病机制及治疗进展[ J]. 中华神经外科疾病研究杂志, 2016, 15(2): 190-192.
WANG Qiuhong, WEI Ruili. Progress in pathogenesis and treatment of traumatic optic nerve injury[ J]. Chinese Journal of Neurosurgical Disease Research, 2016, 15(2): 190-192.
58、Frik J, Merl-Pham J, Plesnila N, et al. Cross-talk between monocyte invasion and astrocyte proliferation regulates scarring in brain ainjury[ J]. EMBO Rep, 2018, 19(5): e45294.Frik J, Merl-Pham J, Plesnila N, et al. Cross-talk between monocyte invasion and astrocyte proliferation regulates scarring in brain ainjury[ J]. EMBO Rep, 2018, 19(5): e45294.
59、Song G, Yang R, Zhang Q, et al. TGF-β Secretion by M2 macrophages induces glial scar formation by activating astrocytes in vitro[ J]. J Mol Neurosci, 2019, 69(2): 324-332.
Song G, Yang R, Zhang Q, et al. TGF-β Secretion by M2 macrophages induces glial scar formation by activating astrocytes in vitro[ J]. J Mol Neurosci, 2019, 69(2): 324-332.
60、Schachtrup C, Ryu JK, Helmrick MJ, et al. Fibrinogen triggers astrocyte scar formation by promoting the availability of active TGF-beta after vascular damage[ J]. J Neurosci, 2010, 30(17): 5843-5854.Schachtrup C, Ryu JK, Helmrick MJ, et al. Fibrinogen triggers astrocyte scar formation by promoting the availability of active TGF-beta after vascular damage[ J]. J Neurosci, 2010, 30(17): 5843-5854.
61、Huang XQ, Zhang XY, Wang XR, et al. Transforming growth factor β1-induced astrocyte migration is mediated in part by activating 5-lipoxygenase and cysteinyl leukotriene receptor 1[J]. J Neuroinflammation, 2012, 9: 145.Huang XQ, Zhang XY, Wang XR, et al. Transforming growth factor β1-induced astrocyte migration is mediated in part by activating 5-lipoxygenase and cysteinyl leukotriene receptor 1[J]. J Neuroinflammation, 2012, 9: 145.
62、Kumar H, Choi H, Jo MJ, et al. Neutrophil elastase inhibition effectively rescued angiopoietin-1 decrease and inhibits glial scar after spinal cord injury[ J]. Acta Neuropathol Commun, 2018, 6(1): 73.Kumar H, Choi H, Jo MJ, et al. Neutrophil elastase inhibition effectively rescued angiopoietin-1 decrease and inhibits glial scar after spinal cord injury[ J]. Acta Neuropathol Commun, 2018, 6(1): 73.
63、Wang SM, Hsu JC, Ko CY, et al. Astrocytic CCAAT/enhancer-binding protein delta contributes to glial scar formation and impairs functional recovery after spinal cord injury[J]. Mol Neurobiol, 2016, 53(9): 5912-5927.Wang SM, Hsu JC, Ko CY, et al. Astrocytic CCAAT/enhancer-binding protein delta contributes to glial scar formation and impairs functional recovery after spinal cord injury[J]. Mol Neurobiol, 2016, 53(9): 5912-5927.
64、孙颖健, 郑雅娟. 干细胞移植治疗视网膜神经节细胞损伤性疾病的研究进展[ J]. 国际眼科杂志, 2015, 15(4): 630-632.
SUN Yinjian, ZHENG Yajuan. Progress in stem cell transplantation for treatment of retinal ganglion cell damage diseases[ J]. International Eye Science, 2015, 15(4): 630-632.孙颖健, 郑雅娟. 干细胞移植治疗视网膜神经节细胞损伤性疾病的研究进展[ J]. 国际眼科杂志, 2015, 15(4): 630-632.
SUN Yinjian, ZHENG Yajuan. Progress in stem cell transplantation for treatment of retinal ganglion cell damage diseases[ J]. International Eye Science, 2015, 15(4): 630-632.
65、Tzameret A, Sher I, Belkin M, et al. Transplantation of human bone marrow mesenchymal stem cells as a thin subretinal layer ameliorates retinal degeneration in a rat model of retinal dystrophy[ J]. Exp Eye Res, 2014, 118: 135-144.Tzameret A, Sher I, Belkin M, et al. Transplantation of human bone marrow mesenchymal stem cells as a thin subretinal layer ameliorates retinal degeneration in a rat model of retinal dystrophy[ J]. Exp Eye Res, 2014, 118: 135-144.
66、Chen M, Xiang Z, Cai J, et al. The anti-apoptotic and neuro-protective effects of human umbilical cord blood mesenchymal stem cells (hUCB-MSCs) on acute optic nerve injury is transient[J]. Brain Res, 2013, 1532: 63-75.Chen M, Xiang Z, Cai J, et al. The anti-apoptotic and neuro-protective effects of human umbilical cord blood mesenchymal stem cells (hUCB-MSCs) on acute optic nerve injury is transient[J]. Brain Res, 2013, 1532: 63-75.
67、Lidgerwood GE, Hewitt AW, Pébay A, et al. Human pluripotent stem cells for the modelling of diseases of the retina and optic nerve: toward a retina in a dish[ J]. Curr Opin Pharmacol, 2019, 48: 114-119.Lidgerwood GE, Hewitt AW, Pébay A, et al. Human pluripotent stem cells for the modelling of diseases of the retina and optic nerve: toward a retina in a dish[ J]. Curr Opin Pharmacol, 2019, 48: 114-119.
68、Luo L, He Y, Jin L, et al. Application of bioactive hydrogels combined with dental pulp stem cells for the repair of large gap peripheral nerve injuries[ J]. Bioact Mater, 2021, 6(3): 638-654.Luo L, He Y, Jin L, et al. Application of bioactive hydrogels combined with dental pulp stem cells for the repair of large gap peripheral nerve injuries[ J]. Bioact Mater, 2021, 6(3): 638-654.
69、Hertz J, Qu B, Hu Y, et al. Survival and integration of developing and progenitor-derived retinal ganglion cells following transplantation[ J].Cell Transplant, 2014, 23(7): 855-872.Hertz J, Qu B, Hu Y, et al. Survival and integration of developing and progenitor-derived retinal ganglion cells following transplantation[ J].Cell Transplant, 2014, 23(7): 855-872.
70、Nakano T, Ando S, Takata N, et al. Self-formation of optic cups and storable stratified neural retina from human ESCs[ J]. Cell Stem Cell,2012, 10(6): 771-785.Nakano T, Ando S, Takata N, et al. Self-formation of optic cups and storable stratified neural retina from human ESCs[ J]. Cell Stem Cell,2012, 10(6): 771-785.
71、Wang LJ, Liu LP, Gu XL, et al. Implantation of adipose-derived stem cells cures the optic nerve injury on rats through inhibiting the expression of inflammation factors in the TLR4 signaling pathway[ J].Eur Rev Med Pharmacol Sci, 2018, 22(5): 1196-1202.Wang LJ, Liu LP, Gu XL, et al. Implantation of adipose-derived stem cells cures the optic nerve injury on rats through inhibiting the expression of inflammation factors in the TLR4 signaling pathway[ J].Eur Rev Med Pharmacol Sci, 2018, 22(5): 1196-1202.
72、Hu ZL, Li N, Wei X, et al. Neuroprotective effects of BDNF and GDNF in intravitreally transplanted mesenchymal stem cells after optic nerve crush in mice[ J]. Int J Ophthalmol, 2017, 10(1): 35-42.Hu ZL, Li N, Wei X, et al. Neuroprotective effects of BDNF and GDNF in intravitreally transplanted mesenchymal stem cells after optic nerve crush in mice[ J]. Int J Ophthalmol, 2017, 10(1): 35-42.
73、Apara A, Goldberg JL, et al. Molecular mechanisms of the suppression of axon regeneration by KLF transcription factors[ J]. Neural Regen Res, 2014, 9(15): 1418-1421.Apara A, Goldberg JL, et al. Molecular mechanisms of the suppression of axon regeneration by KLF transcription factors[ J]. Neural Regen Res, 2014, 9(15): 1418-1421.
74、Li S, Yang C, Zhang L, et al. Promoting axon regeneration in the adult CNS by modulation of the melanopsin/GPCR signaling[ J]. Proc Natl Acad Sci U S A, 2016, 113(7): 1937-1942.Li S, Yang C, Zhang L, et al. Promoting axon regeneration in the adult CNS by modulation of the melanopsin/GPCR signaling[ J]. Proc Natl Acad Sci U S A, 2016, 113(7): 1937-1942.
75、Park KK, Liu K, Hu Y, et al. Promoting axon regeneration in the adult CNS by modulation of the PTEN/mTOR pathway[ J]. Science, 2008,322(5903): 963-966.Park KK, Liu K, Hu Y, et al. Promoting axon regeneration in the adult CNS by modulation of the PTEN/mTOR pathway[ J]. Science, 2008,322(5903): 963-966.
76、Chiha W, Bartlett CA, Petratos S, et al. Intravitreal application of AAV-BDNF or mutant AAV-CRMP2 protects retinal ganglion cells and stabilizes axons and myelin after partial optic nerve injury[ J]. Exp Neurol, 2020, 326: 113167.Chiha W, Bartlett CA, Petratos S, et al. Intravitreal application of AAV-BDNF or mutant AAV-CRMP2 protects retinal ganglion cells and stabilizes axons and myelin after partial optic nerve injury[ J]. Exp Neurol, 2020, 326: 113167.
77、Moore DL, Blackmore MG, Hu Y, et al. KLF family members regulate intrinsic axon regeneration ability[J]. Science, 2009,326(5950): 298-301.Moore DL, Blackmore MG, Hu Y, et al. KLF family members regulate intrinsic axon regeneration ability[J]. Science, 2009,326(5950): 298-301.
78、Qin S, Zou Y, Zhang CL, et al. Cross-talk between KLF4 and STAT3 regulates axon regeneration[ J]. Nat Commun, 2013, 4: 2633.Qin S, Zou Y, Zhang CL, et al. Cross-talk between KLF4 and STAT3 regulates axon regeneration[ J]. Nat Commun, 2013, 4: 2633.
79、Pernet V, Joly S, Dalkara D, et al. Long-distance axonal regeneration induced by CNTF gene transfer is impaired by axonal misguidance in the injured adult optic nerve[ J]. Neurobiol Dis, 2013, 51: 202-213.Pernet V, Joly S, Dalkara D, et al. Long-distance axonal regeneration induced by CNTF gene transfer is impaired by axonal misguidance in the injured adult optic nerve[ J]. Neurobiol Dis, 2013, 51: 202-213.
80、Borgens RB, Roederer E, Cohen MJ, et al. Enhanced spinal cord regeneration in lamprey by applied electric fields[ J]. Science, 1981,213(4508): 611-617.Borgens RB, Roederer E, Cohen MJ, et al. Enhanced spinal cord regeneration in lamprey by applied electric fields[ J]. Science, 1981,213(4508): 611-617.
81、Gokoffski KK, Jia X, Shvarts D, et al. Physiologic electrical fields direct retinal ganglion cell axon growth in vitro[ J]. Invest Ophthalmol Vis Sci,2019, 60(10): 3659-3668.Gokoffski KK, Jia X, Shvarts D, et al. Physiologic electrical fields direct retinal ganglion cell axon growth in vitro[ J]. Invest Ophthalmol Vis Sci,2019, 60(10): 3659-3668.
82、Ellis-Behnke RG, Liang YX, You SW, et al. Nano neuro knitting:peptide nanofiber scaffold for brain repair and axon regeneration with functional return of vision[ J]. Proc Natl Acad Sci U S A, 2006,103(13): 5054-5059.Ellis-Behnke RG, Liang YX, You SW, et al. Nano neuro knitting:peptide nanofiber scaffold for brain repair and axon regeneration with functional return of vision[ J]. Proc Natl Acad Sci U S A, 2006,103(13): 5054-5059.
83、Yang TC, Chuang JH, Buddhakosai W, et al. Elongation of axon extension for human iPSC-derived retinal ganglion cells by a nano-imprinted scaffold[ J]. Int J Mol Sci, 2017, 18(9): 2013.Yang TC, Chuang JH, Buddhakosai W, et al. Elongation of axon extension for human iPSC-derived retinal ganglion cells by a nano-imprinted scaffold[ J]. Int J Mol Sci, 2017, 18(9): 2013.
84、Bei F, Lee HHC, Liu X, et al. Restoration of visual function by enhancing conduction in regenerated axons[ J]. Cell, 2016, 164(1/2): 219-232.Bei F, Lee HHC, Liu X, et al. Restoration of visual function by enhancing conduction in regenerated axons[ J]. Cell, 2016, 164(1/2): 219-232.
85、Lim JH, Stafford BK, Nguyen PL, et al. Neural activity promotes long-distance, target-specific regeneration of adult retinal axons[ J]. Nat Neurosci, 2016, 19(8): 1073-1084.Lim JH, Stafford BK, Nguyen PL, et al. Neural activity promotes long-distance, target-specific regeneration of adult retinal axons[ J]. Nat Neurosci, 2016, 19(8): 1073-1084.
86、Gordon K, Del Medico A, Sander I, et al. Gene therapies in ophthalmic disease[ J]. Nat Rev Drug Discov, 2019, 18(6): 415-416.Gordon K, Del Medico A, Sander I, et al. Gene therapies in ophthalmic disease[ J]. Nat Rev Drug Discov, 2019, 18(6): 415-416.