This study reviewed all the patients who had undergone
consecutive intravenous FFA from 29 March 2010 to 29
February 2012 in the Department of Ophthalmology,
Guangdong General Hospital, China (when patients had
multiple FFAs within the period, only the first procedure
was included). Patients who reported any allergic history
and presence of atopy, patients who had any prior reaction
which was not a severe allergic AR and patients who
complained of asymptomatic hypertension and any renal
problem with serum creatinine levels lower than 250 µmol/L
or with which patients had been undergoing renal dialysis
were not excluded from FFA. No patient was excluded on
the ground of age either.
Information in the following was obtained prior to the
procedure: informed consent, age, gender, ethnic group, daily
smoking habits, weekly alcohol intake, prior FFAs, medication,
allergic history, past medical history and actual discomfort.
As a routine, blood pressure was measured before the
procedure. Steroids or antihistamines were prescribed
before FFA only when patients had a need for an ocular
or systemic disease and when patients were afraid of
the allergic reactions to fluorescein. The patient’s eyes
were dilated using 0.5% tropicamide and phenylephrine
compound eye drop. Fifteen minutes after a pre-injection
of 3 mL of 1‰ sodium fluorescein, 3 mL or 10 mg/kg of
20% sodium fluorescein (Guangzhou Baiyun Shan Ming
Xing Pharmaceutical Co. Ltd., Guangzhou, China) was
injected in each patient around eight seconds. All patients
were told to inform the participating doctor and nurse if
they felt unwell and the doctor also reviewed each patient to
see whether they suffered any discomfort during and after
the procedure. Reactions were divided into mild, moderate
and severe reaction and death according to the classification
suggested by Yannuzzi et al.
(13).
Chi-square test was used to find associations between
the categorical variables. P<0.05 was considered statistically
significant. The correlations between all variables and
ARs were evaluated by multivariate logistic regression
analysis. Odd ratio (OR) and confidence interval (CI) were
calculated.
Results
As a result, a total of 829 patients who were Asian were
obtained: 459 were male with a mean age of 54.5±16.6 years
and 370 were female with a mean age of 56.7±16.2 years.
No statistically significant difference was found between the
proportions of males and females (
P=0.1566). Ages ranged
from four to ninety.
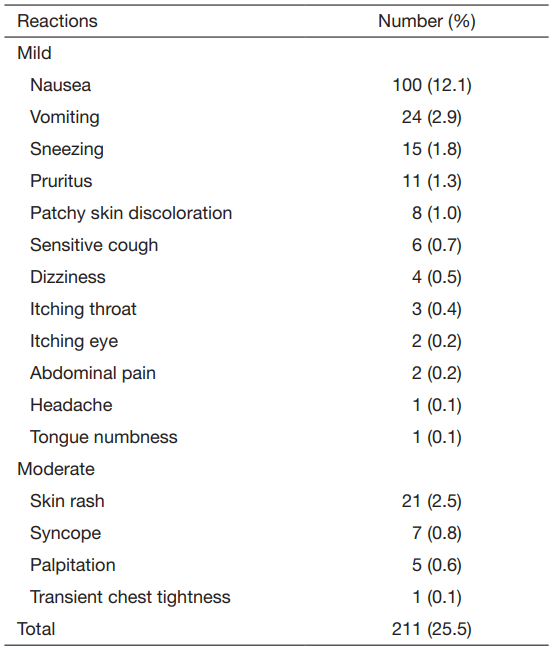
ARs occurred in 184 patients (22.2%) and the majority
of reactions were nausea (Table 1). Twenty-four patients
had two or more reactions. There were no cases of severe
ARs and death. All sensitive reactions of sneezing, sensitive
cough, itching eye or throat happened in patients without
the outbreak of hay fever. Eleven (52.4%) of 21 patients
with prior reactions experienced similar reactions again
(6 of them were emetic reactions, 2 fixed drug eruption,
2 urticaria and 1 sneezing). Seven (46.7%) of 15 patients
who experienced motion sickness on the way to the clinic
for the procedure experienced emetic reactions during FFA.
Table 1 Different types of adverse reactions (n=829)
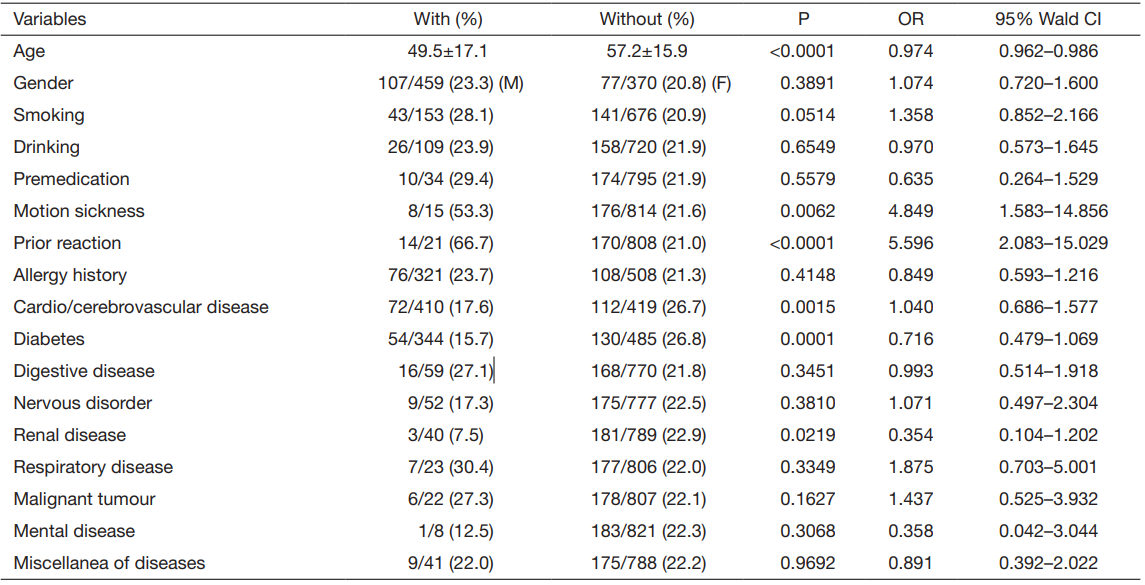
When the relationships between ARs and variables of
age, gender, smoking, drinking, premedication of steroids
or antihistamines, prior reactions, motion sickness, and a
history of allergy, cardio/cerebrovascular disease, diabetes,
digestive disease including liver problems, nervous
disorder, renal disease, respiratory disease, malignant tumour, mental disease and miscellanea of diseases were
evaluated separately, the mean age of patients with ARs
was statistically significant younger than that of those
without ARs (
P<0.0001), and significant higher risk of ARs
were found in patients with motion sickness (
P=0.0062)
and a history of prior reaction (
P<0.0001), interestingly,
lower risk were found in patients with a history of cardio/
cerebrovascular disease (
P=0.0015), diabetes (
P=0.0001),
and renal disease (
P=0.0219); however, when all these
variables were evaluated simultaneously with the presence of
ARs by multivariate logistic regression analysis, statistically
significant correlations with ARs were only revealed in age
(OR 0.974; 95% CI, 0.962–0.986), motion sickness (OR
4.849; 95% CI, 1.583–14.856) and prior reaction (OR
5.596; 95% CI, 2.083–15.029), but a history of cardio/
cerebrovascular disease, diabetes and renal disease (Table 2).
Table 2 Frequency of adverse reactions (mean age) and multivariate logistic regression analysis result (n=829)
M, males; F, females; OR, odd ratio; CI, confidence interval.
In addition, a strong statistically significant decreasing
tendency of emetic reaction along with increasing age is
shown in Table 3 (
P<0.0001).
Table 3 Frequency of emetic reactions related to age (n=829)
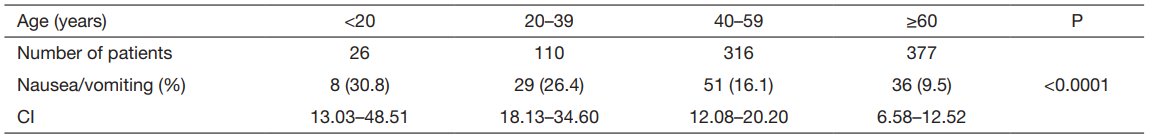
CI, confidence interval.
Discussion
Many factors are suggested related to ARs of FFA. For
evaluating the effects of these factors, multivariate analysis may be the best way to avoid influence from each other.
To our knowledge, risk factors educed from a multivariate
analysis has never been addressed. This study demonstrated
only age, prior reactions and motion sickness but gender,
premedication, smoking and drinking habits, and a history
of allergy and a series of systemic diseases correlated with
ARs in the multivariate logistic regression analysis.
The
average age of patients with ARs was significantly younger
than that of those without ARs. Therefore, the proportion
of young patients, patients with prior reaction and patients
with motion sickness must affect the overall rate of ARs in
a subgroup. That is why an obviously low frequency of ARs
in patients with cardio/cerebrovascular diseases, diabetes
and renal diseases did not reach the level of statistical
significance in the multivariate logistic regression analysis
in this study because these diseases usually happen in
older people. The lower risks to FFA for these patients in
the univariate analysis which had ignored the age factor
obviously were false positive. Also because all of these
variables are not taken into account in the previous studies,
it is not surprising why the reported outcomes on single
possible causative factor are widely inconsistent.
Emetic reaction which is part of vasovagal reaction is the
main AR during FFA. Vasovagal susceptibility is probably
present in all healthy humans but may vary in genetic
basis (5). During FFA patients with prior emetic reactions
have been presented more like to be nauseous and vomit in
previous and our studies (4,5). Vasovagal reactions also may
be precipitated by many factors such as fear, severe pain,
instrumentation and dehydration. So young people who are
more ready for developing dehydration and generally more
scared of injection and examinations would be more likely
to develop emetic reaction. In fact, the frequency of emetic
reaction was observed reduced alone with increasing age in
this study. Musa et al. also found adverse events in the group
aged ≥90 are less than that in the 70- to 79-year-old group,
unfortunately, they did not find a significantly difference in
ARs between those aged ≥75 years and aged <75 years (8).
It may be owing to a higher proportion of patients with
prior reactions in the older group. Motion sickness, which is
induced by nystagmus belonging to an oculo-emetic reflex,
certainly would aggravate its impact on emetic episodes
during FFA. However, patients only with a history of motion
sickness before are not more likely to be nauseous (5).
In addition, chronic consumption of exogenous toxins is
suggested to increase tolerance to emetogenic stimulations (15,16). Yet, no statistical correlation between smoking or
drinking and adverse events was found in our study. The
rates of nausea in the groups with the habits of smoking
(12.4%) and drinking (13.8%) were in fact comparable with
those in the groups without the habits (12.0% and 11.8%
respectively).
In clinical practice, histories of allergy and severe diseases
such as myocardial infarction and malignant tumour are
generally viewed as a relative contraindication for FFA. Yet,
until now only the relationship between ARs and a history of
allergy, ischaemic heart disease including hypertension and
diabetes has been discussed. Our result identified the gap
in the literature in that the relationship between ARs and a
history of other diseases has not been delivered. Fluorescein
is excreted mainly in the urine unchanged and partly by way
of liver by conjugation with glucuronide and is not directly
hepatotoxic (17). Hence, FFA is relatively safe for patients
with renal and liver insufficiency when their estimated
glomerular filtration rate is 45 mL/min/1.73 m2
and greater
or they have undergone renal dialysis
although until now
there is no satisfactory method to estimate the degree of
risk associated with drug in patients with chronic liver
disease (18). Our study demonstrated a history of renal or
liver problem and malignant tumour did not increase adverse
events. Despite the result from Lira et al. that patients with
a history of allergy, hypertension and diabetes have a higher
occurrence of ARs, but which mainly are mild type, Musa et al.
showed ischaemic heart disease does not correlate with
ARs and patients with systolic blood pressure ≥160 mmHg
(1 mmHg =0.133 kPa) even have a significantly lower reaction
rate than those with blood pressure <160 mmHg (8,9).
Kalogeromitros et al. revealed atopy does not increase the ARs risk either (14). In our study a history of cardio/
cerebrovascular diseases, diabetes and allergy also was
not correlated with ARs. It may be explained by the fact
that blood pressure regulatory genes could be oppositely
affected in vasovagal reaction and hypertension and an
existing allergic condition and an atopic family history do
not increase the risk of an allergic reaction to a drug (19-21).
Moreover, no ST-T changes or rhythm disturbances are
displayed on electrocardiograms in patients including those
with diabetes, cardiac diseases and other systemic diseases
during FFA (22). In addition, histories of digestive disease
and mental disease suggest prone to lower threshold of
emetic reaction and anxiety. However, these patients did
not exhibit a higher rate of ARs in this study. It is therefore
reasonable to assume that FFA is quite safe for a relatively
healthy patient no matter what a past medical history and
allergy history he has.
The overall rate of ARs is estimated from 0.6% to 22% (3,4,14,23,24). Most of ARs are mild. The prevalence of
nausea varies widely from 0.7% to 15% (3,4,11,14,24). And
the main moderate AR keeps on skin rash which occurs
from 0.2% to 2.0% (3,8,9,13). Our results of 25.5%, 12.1%
and 2.5% of the overall ARs, nausea and skin rash were at
the high end of or beyond previous ranges. Asian ethnic
group, pre-injection of 1‰ sodium fluorescein, a relatively
higher size of young patients and patients with prior
ARs, the physician’s careful observation of any minimum
clinical abnormality that is not reported by patients and
the conventional review by asking patients whether they
experience side effects that are not readily observable were
suggested to contribute to the higher rates (5,13).
In conclusion, FFA is a relatively safe procedure. A
history of allergy and severe systemic diseases which are
relatively under controlled should not be viewed as a
contraindication. The most common AR, emetic reaction,
is correlated with age, prior reactions and motion sickness.
These risk factors should be considered in future study on
ARs to the dye. Multivariable analysis is also recommended
to assess risk factors.