Clinical research data management includes verifying the
completeness and correctness of the data collection in
clinical trials, ensuring the support of experimental data
by statistical analyses, and illustrating and interpreting
the results of experiments, all throughout the clinical
research process. Traditionally, clinical data management
has an internationally recognized general procedure
(1),
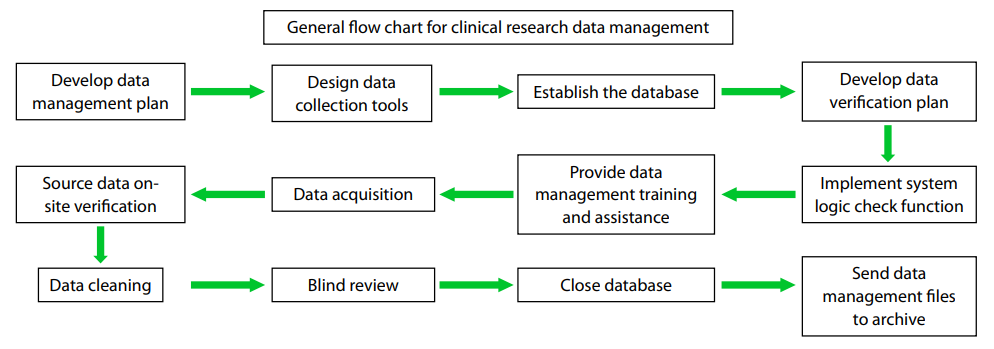
as summarized in Figure 1. Currently, clinical trial data
acquisition methods are mainly classified as one of two types, i.e., paper data capture (PDC) and electronic data
capture (EDC ). Overall, EDC has the advantages of
improving test efficiency and data quality, reducing costs,
shortening the study period, decreasing loss, and enhancing
the user’s experience; the development and application
of EDC tools in the field of clinical research have greatly
promoted the process of digitalization in clinical data
management.
Figure 1 General flowchart for clinical research data management.
A database is an advanced stage of data management and
distinctively different from the traditional data management
approaches mainly in the following aspects: first, databases
make the data independent of applications, thereby
enabling centralized data management and improving
the efficiency of data through data sharing and reducing
data redundancy; second, they establish links between
different databases so that the connection between real world
information is reflected; therefore, the database is no
longer just a simple tool for data collection
(2). According
to its nature, a database can be divided into one of three
categories: literature databases, numerical databases, and
factual databases, each of which can be further subdivided.
Literature databases include professional literature
databases, library bibliographic databases, combined catalog
databases, patent literature databases, full-text databases,
and others. Numeric databases include scientific databases,
engineering and technology databases, and others. Factual
databases include economic and business databases,
management databases, public services databases, and
others
(3). All three main categories of databases have been
widely used in clinical studies.
Based on the complexity of the data model, database
technology can be accordingly divided into three stages,
i.e., the first-generation nest and hierarchical database
system, the second-generation relational database system,
and the third-generation database system mainly featuring object-oriented models. With the increasing data types
and complexity of the data association, nested and layered
databases solely relying on the tree-root node styled
structure have been unable to meet the growing demand
for database functions and have been mostly discarded.
At present, the most popular structure is the second generation
relational database, consisting of relational data
structures. This type of database classifies, merges, and
selects data through transforming the data structure into a
two-dimension relational form and builds data platforms
with complex and comprehensive management functions.
However, different applications have specific functional
requirements for databases that cannot be completely
covered by relational databases. Therefore, the third generation
databases, with richer data models and more
personalized functions of management and analysis, have
emerged but have only been applied to a small number of
fields, such as military applications, because of the limitation
of their high cost
(2).
Three factors are critical to successful database
construction: management, data, and technology. First,
management is the foundation of database construction,
which includes decisions in the early stages and the
formulation of corresponding policies, strategies, and
measures; software development management, data
acquisition, and quality control surrounding software
engineering in the development stage; the security,
confidentiality, updating, and restructuring of data; and the
three types of maintenance management for application
software (corrective maintenance, adaptive maintenance,
and improvement maintenance) in the stages of service
and maintenance. Second, data are the basis of database
construction, and the reliability and stability of the data
source are the prerequisites for everything else associated
with the database, so stricter requirements for data collection, entry, and processing have been proposed.
Lastly, technology is the most direct security for database
construction and has wide and complex applications, mainly
including database technology, computer technology, and
network and communication technologies
(4).
Construction of clinical databases and
procedures for data entry, review, and
management
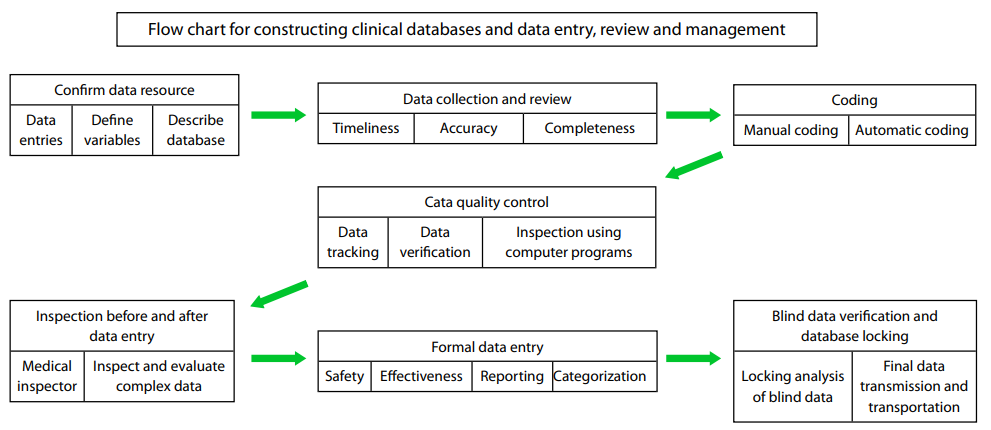
Database construction, data entry, review, and management
are very complex and meticulous processes, as shown in
Figure 2. However, in actual operations, attention should be
paid to the following aspects: (I) verifying the source of data,
which mainly includes data from the site of the research and
the associated laboratories. Then, the data to be collected
(i.e., the research variables), the definitions of the variables
(i.e., definitions for concept and operation), the description
of the database (i.e., the data type, single or replicate), a
confirmatory note on the consistency between the data and
the raw data, and a confirmatory note on data validation
(i.e., inspections on the values and the data range, missing
values, and logic checks) are all entered; (II) collecting and
reviewing the data, which requires timeliness, completeness,
and accuracy. Specifically, first, “timeliness” is to ensure
the quality of the raw data and to reduce the time required
for the subsequent review; “completeness” is to require
the collection of all the data on the subject; and “accuracy”
is to require that first, the designed case report should
have better operability, quantitative indicators should be
adopted as much as possible, and appropriate quantification
should also be applied to the soft indicators. Second,
the laboratory conditions and operators should be kept
relatively constant. Third, the clinicians or data collectors
who collect the data or fill out the case report forms must
be subjected to certain training. Fourth, recoding data from
the raw data should be minimized; in case of recoding,
careful reviews must be performed to ensure consistency
between the case report forms and the original data. After
completing the data collection, data review is necessary,
which includes self-review and supervision and inspection;
(III) coding, which includes manual coding and automatic
coding techniques including dictionaries (MedDRA,
WHO-ART, ICD, COSTART ); (IV) data quality control.
Commonly used methods include data tracking, data entry
and validation, data verification, data questionnaires, checks
using computer programs, and others; (V) data verification
before and after data entry. Clinical data are verified by
qualified medical inspectors; verification includes examining
and assessing complex clinical data to find subtle difference
in the data, after which data managers perform quality
control on the evaluation; ( VI) formal data entry and
database construction. The requirements for data entry
are either that problematic data should be resolved and the
entry and verification of major data should be complete
or the data have been sampled for quality control, and the
major data related to safety and effectiveness have been
evaluated with quality control checks, and all of the events
that are not in accordance with research programs have
been reported, categorized, and clarified with the related
effects. After the entry has been completed and has met
the requirements, data collation can be performed, and the
data can be transmitted to the final database; (VII) blind
data verification and database locking. After confirming the authenticity of the information and completing the blind
data verification, the database is locked, and the process
proceeds to the final analysis; the objectives and order of the
analysis are further clarified together with the researchers.
Then, the database is accepted, and a preliminary analysis
on the information is conducted, which is followed by a
preliminary data analysis to develop the draft report for
the last analysis or revised statistical analysis plan. After
verification, the database should be locked to prevent misuse
and unauthorized modification, and a list of data required
by the clinical study report should be generated. Lastly, the
data are transmitted and archived, in which the data are
sent to sponsors, statisticians, and compliance supervising
departments
(5).
Figure 2 Flowchart for constructing clinical databases and data entry, review and management.
The important role for the clinical database on
determining the level of clinical research
The important role for the clinical database on determining
the level of clinical research is mainly reflected in the
following aspects: first, a clinical database is a prerequisite
for data analysis and reaching a conclusion; second, a clinical
database is one of the measurement bases for the level of
research. The huge amount of data generated in a clinical
study can greatly improve both the efficiency of research
and the value of data through regularizing the database, and
it can provide lessons and bases for researchers in the long term
process of the study. Analyzing the database can test
the clinical study and reveal significance as well as determine
the authenticity and reliability of the clinical study, thus
effectively regulating the design and implementation of the
clinical study (6). The data in clinical research are collected
and managed in real-time, and any data management
mistakes may break the original tightly interlocking links in
the clinical trial, resulting in a loss that cannot be reversed
afterward. The quality of the data management reflects
the implementation status of the experimental design and
scheme by the investigator in the clinical trial, while alluding
to the scientifi c approach and knowledge level of the clinical
researchers. Data management is also often subjected to the
largest part of the audit of the implementation of clinical
research and is one of the major responsibilities of supervisors
and inspectors (7).
Effect of the reanalysis of RCTs on the
conclusions of a study
The reanalysis of RCTs is one of the latest developments for
databases applied to clinical research. In 2014, the Journal
of the American Medical Association published a landmark
study on the reanalysis of RCTs, in which the investigators
found that in 37 cases, only five authors who reanalyzed
RCTs were totally unrelated to the initial experiments.
In terms of the experimental method, in the 37 cases,
46 different methods, including statistics and analysis
methods, were applied, and in the RCT reanalysis results,
35% of the reanalysis drew different conclusions about
patient treatment method from the initial experiments (8).
This experiment representatively indicated that the
reanalysis of RCTs on data from existing clinical studies
is feasible and can come to either the same or different
conclusions as the original trial. The role of the reanalysis is
to examine and supplement the initial experimental results
or to find loopholes and errors. However, this analysis
method still has some risks that have sparked disputes in
academia because of its short history. First, the reanalysis of
RCTs may pose a potential safety threat to patient privacy,
resulting in the exposure of patient medical information.
Second, inappropriate mining on data sets has occurred
in the reanalysis of RCTs. Third, forged and false results
have emerged in some reanalysis of RCTs. Fourth, some
reanalysis of RCTs led to the leak of trade secrets
(9).
Currently, only a limited number of reanalysis of RCTs
have been reported in the literature, and the investigators
of the reanalysis and the methods that were adopted all
affected the results of the analysis
(8,9). Therefore, at
present, the quality and credibility of the reanalysis of
RCTs are worrisome, and it is imperative to promulgate
standards of data sharing and RCT reanalysis that are more
authoritative and have a broader influence. Only after a
unified and authoritative standard is developed will it be
possible to mitigate various issues such as the violation of
patient privacy, trade secret leaks, or experiments performed
to advance a particular agenda, among others, so that RCT
reanalysis can be as effective as possible.
The development modes of cloud-based clinical
research databases
Origin of cloud computing
After years of development, database technology has
gradually matured and is playing an important role in
today’s information society. However, it has also exposed
some problems with its application, mainly in two aspects.
First, there is an issue with updating data. Traditional databases lack real-time and initiated updates. Because there
are mostly historical data in traditional data warehouses and
the data retrieving cycle generally lasts several days or even
a week, it is difficult to process data in real time. Meanwhile,
traditional data warehouses use Extraction Transformation
Loading (ETL) and adopt periodic batch updates in which
the time and data of the update are preset and unable to
change as needed. Second, the application scope and field of
traditional data warehouses are rather narrow (10).
In recent years, the application of cloud computing in
databases has resulted in new advances such as data clouds,
real-time data, intelligent analysis of data, and others,
providing new ideas for solving the above problems. Socalled
cloud computing refers to network technology that
uses a computer as a carrier and the network and the Internet
as bases to provide real-time services to users. This is an adhoc
service, i.e., it provides what the user needs and charges
service fees based on the user’s usage, which greatly improves
the rate of resource sharing (11). The essence of a cloud
computing-based database is a large-scale centralized joint
database management system, distributed across physical
structures but logically belonging to the same system. The
combination of cloud computing and databases has two forms:
databases running in the cloud and cloud databases (12).
The development of these two forms of databases is still in
its infancy; while evolving in their own ways, they also bring
more possibilities to database applications.