
Purpose: To investigate the effect of intravitreal injection of basic fibroblast growth factor (bFGF) on activation and proliferation of endogenous retinal progenitor cells in the Royal College of Surgeons (RCS) rat.
Methods: Twenty-four rats were studied after the 30th postnatal day (≥30). Eighteen RCS-p+/LAV rats were divided into 3 groups: bFGF-treated, vehicle-treated, and untreated groups randomly, and 6 RCS-ray+p+/Lav rats were used as normal controls. 6 μl of bFGF (5 μg /10 μl) or vehicle was injected into the vitreous on day 31, 33, and 35 after birth (P31, P33, P35) in the bFGF group and vehicle group respectively, and no injections were administered in the untreated and control groups. All the rats were euthanized, and their eyes were enucleated, hemisected, and fixed at 50 days after birth for immunohistochemistry and measurement of outer nuclear layer thickness.
Results: Nestin and Chx10 were positive in all retinal layers. Intravitreal injection of bFGF in retina-dystrophic RCS (RCS-p+/Lav) rats induced intense labeling for the retinal progenitor cell markers Chx10 and Nestin, which were highly colocalized. Fluorescence intensity for both labels was somewhat less in the control rats, and much less in the vehicle-injected rats as well as in the untreated RCS rats. The outer nuclear layer (ONL) was significantly thicker in the bFGF group than that in the vehicle-treated or untreated group (P < 0.01), but thinner than that of the control group (P < 0.01). No significant difference was observed in the ONL thickness between the vehicle group and untreated group (P > 0.05).
Conclusion: bFGF may contribute to the activation of retinal progenitor cells in RCS rats, thus counteracting degeneration by promoting the proliferation of the progenitor cells.
Photoreceptor degeneration due to either intrinsic genetic defects involving the photoreceptors themselves or problems affecting the cells with which the photoreceptors interact presents as a major group of neurodegenerative diseases and is a leading cause of blindness in developed countries. It comprises retinitis pigmentosa, age-related macular degeneration, and a number of associated diseases which collectively account for visual impairment in millions of people in the world [1], whereas there are few clinical treatment options for these conditions. However, studies on animal models with similar patterns of photoreceptor loss have identified the therapeutic potential of several novel approaches directed at limiting the course of photoreceptor loss, including growth factor or neurotrophic factor injection [2-5], gene therapy [6-8], and cell-based therapy [9-10].
Gene therapy is not ready for clinical applications at present [8]. For cell-based therapy to be viable in translation to the clinic, there are several critical requirements to consider. They must be effective in preserving visual function, the cells must be easily obtainable from an ethical cell source, and be available in significant numbers and ideally be in a form that requires little preparation or multiple surgeries. Finally, such cells should be well-characterized, free of identifiable pathogens, demonstrate a normal karyotype through many passages, and ideally be non-immunogenic, so avoiding potential safety concerns. For well-known reasons, it is very difficult for allografts to satisfy these requirements at present [10].
One form of therapy that has attracted substantial attention involves the application of endogenous retinal stem cells, which may satisfy these requirements. In the eyes of mice, retinal stem cells can proliferate clonally in vitro to form spherical colonies of cells (neurospheres) in the presence of basic fibroblast growth factor (bFGF), which can subsequently differentiate into retina-specific cell types including rod photoreceptors, bipolar neurons, and Müller glia [11-12]. Such stem cells are especially interesting because of their potential for regenerating tissues of many kinds and in particular for replacing retinal cells that have been lost in degenerative disease [12].
The adult human retina contains neural progenitor cells, which may have the potential to replace lost photoreceptors and other retinal neurons [13]. This has implications for the pathogenesis and treatment of retinal disorders and degenerations, including retinitis pigmentosa, which is one of the major causes of blindness due to gene mutations that cause photoreceptor degeneration. A puzzling question is why the neural progenitor cells fail to be activated and thus replace degenerated photoreceptors in retinitis pigmentosa. Answers may be found in the neurotrophins, such as bFGF, ciliary neurotrophic factor (CNTF), and brain-derived neurotrophic factor (BDNF), which have been implicated in the proliferation, commitment, differentiation, and survival of retinal progenitor cells [14]. Thus, our hypothesis is that neurotrophins such as bFGF are essential for activation and proliferation of retinal progenitor cells, so that administration of bFGF may promote survival and repair of degenerating retinas by stimulating activation and proliferation of intrinsic retinal stem cells.
The RCS rat is a widely studied animal model of retinitis pigmentosa, in which the inability of the retinal pigment epithelium (RPE) to phagocytize shed photoreceptor outer segments leads to a progressive loss of rod and cone photoreceptors. Since species differences are minimal, and the characteristics of mouse and human progenitor cells are remarkably conserved [15], results from RCS rats are expected to be highly relevant for human therapy. In this study, we addressed whether endogenous retinal progenitor cells were activated to proliferate and produce new neurons in vivo when bFGF was injected intravitreally for therapeutic intervention in the early stages of retinal degeneration in the Royal College of Surgeons (RCS) rat.
The congenic strains of RCS rats available to us were: RCS/Lav (inbred, pink-eyed, dystrophic rats); RCS-rdy+/Lav (congenic, pink-eyed, wild-type, non-dystrophic rats); RCS-p+/Lav (congenic, pigmented, dystrophic rats); and RCS-rdy+p+/Lav (congenic, pigmented, wild-type, non-dystrophic rats). In this study, we chose 18 RCS-p+/Lav rats as experimental animals and 6 RCS-rdy+p+/Lav as unaffected wild-type controls.
Rats were housed, 4 per wire-bottom cage, under controlled temperature (20-25 °C), humidity (40%-70%), and lighting (12h light-dark cycle). All animal procedures were in strict compliance with the ARVO Statement for animal experimentation and were approved by the Animal Care Committee of Sun Yat-sen University Research Centre.
In the bFGF group, bFGF (human recombinant bFGF, PeproTech Inc., Rocky Hill, NJ, purchased from Jinan Biotechnology Co., Jinan University, China) was injected into the vitreous on day 31, 33, and 35 after birth (P31, P33 & P35). In the vehicle group, vehicle alone (without bFGF) was injected at the same times. In the untreated and normal control groups, no injection was performed. Details of injections are given below.
All the rats were euthanized on day 50. Eyes were enucleated and prepared further for immunohistochemical and pathological observation according to the following steps (Fig. 1).
Sections were washed three times for 5 min each with PBS, incubated for 15 min in PBS + 0.1% BSA at 37 °C, washed again three times for 5 min each with PBS, and incubated overnight with primary antibody (mouse anti-rat Nestin antibody and rabbit anti-rat Chx10 antibody, Chemicon International, Inc., Temecula, CA, USA) diluted in PBS at 4 °C. Slides were washed three times for 5 min each in PBS and then incubated with fluorescent secondary antibody (FITC-conjugated goat anti-rabbit IgG, Boster Biological Technology, Ltd., Wuhan, China) for 1 h in a humidified chamber at room temperature. Slides were washed four times for 5 min each in cold PBS and mounted on cover slips in 50% glycerol. After that, slides were observed immediately under a fluorescence microscope (ConfoCor2, Zeiss Ltd. Co., Jena, Germany) and all images were captured.
The structure of the photoreceptor layer was observed with a light microscope. In each hemisphere, the thickness of the ONL was measured at 480 μm intervals at 4 defined points: at 12:00, 3:00, 6:00, and 9:00 1 mm from the optic nerve head. As measurement of ONL thickness in μm alone can be confounded by tilt of the section plane away from the vertical, besides the expression of ONL thickness in μm, we presented the data in terms of numbers of nuclear lamina, which will correct for any undetected tilting of the section plane. Thus, we expressed the data of ONL thickness in both μm and numbers of nuclear lamina.
SPSS 13.0 software was used to compare the difference among groups. A value was considered significant when P < 0.05.
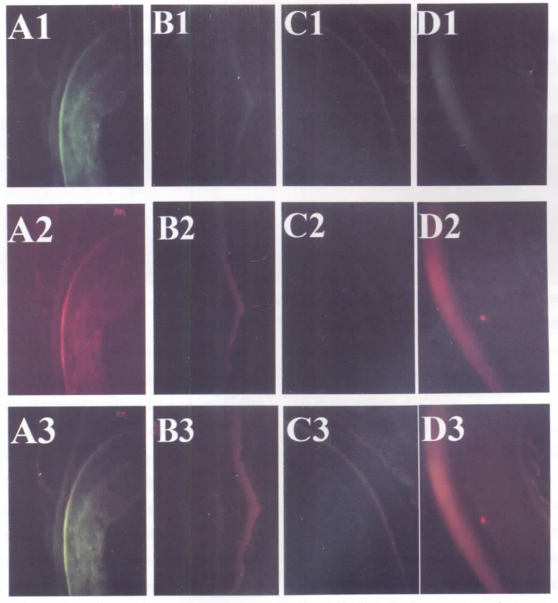
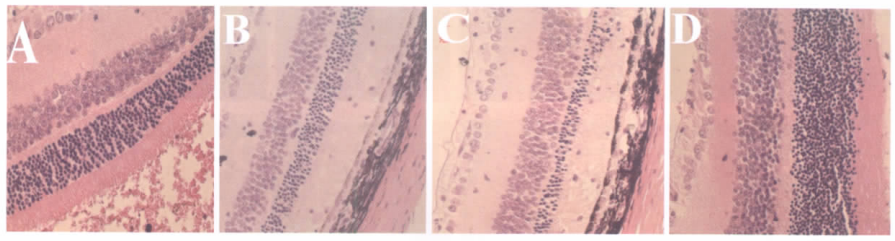
Fig. 3 Retinal histology showed the ONl, thickness of four groups
In spite of the distinction in regenerative potential between stem cells and progenitor cells, their cell-surface markers (surface proteins that are unique to certain cell types, and capable of detection by antibodies or other detection methods) are largely the same. Nestin, an intermediate filament protein expressed by neural progenitors, is a specific biological marker for progenitor cells, since it is found uniquely in all neural progenitors including those in the retina. Chx10 is a specific marker of nuclei in a subpopulation of retinal progenitor cells and is expressed in divided retinal progenitor cells early in development. The co-expression of Nestin and Chx10 in the retina means the existence of retinal progenitor cells.
bFGF (FGF2) is a protein with 146 amino acids and relative molecular mass 16,500 [16]. In vitro, it increased the proportion of progenitor cells forming clonal primary spheres, and the presence of FGF-2-responsive adult human retinal progenitor cells that divide, express Nestin, produce neurospheres, and differentiate to express markers specific to neuronal, photoreceptor, or glial lineage by 4 times [12-13].
In the present experiment, it showed that intravitreal injection of basic fibroblast growth factor (bFGF) induced increases in immunofluorescence for Chx10 and Nestin, while injection of vehicle alone had little effect. This suggested that bFGF can stimulate endogenous retinal progenitors to self-proliferate and migrate into the central retina. The retina in the control group, at the same age as the other three groups, was well-developed and had no need for retinal progenitor cells to activate and proliferate, so the expression of Chx10 and Nestin was stronger than the vehicle and untreated groups but weaker than the bFGF group. On the other hand, intravitreal injection of bFGF induced an increase in ONL thickness and structural stabilization of photoreceptors, suggesting that bFGF could stimulate endogenous retinal progenitors to differentiate into photoreceptors. Of course, other mechanisms of bFGF may also play a role in maintaining a thick ONL, for example, inhibition of photoreceptor apoptosis [17].
Retina is a well-characterized structure, which is arranged in a stereotypical laminar organization. Neural progenitors with retinal potential are present in the ciliary body epithelium [11, 18], the iris pigment epithelium [19-20], and the peripheral margin of the postnatal retina [17, 21]. In this study, the site at which bFGF stimulated retinal progenitor cells to reactivate, proliferate, and differentiate was presumed to be in the peripheral retinal margin, since in mammals the iris pigment epithelium is separated from the vitreous body by aqueous humor and the ciliary body is a non-permissive environment for cell migration and neurogenesis [22].
bFGF has well-documented protective neurotrophic activity in rats with photoreceptor degeneration [23-24]. However, although intravitreal injection of bFGF delays photoreceptor degeneration (as in the present experiment), such treatment does not allow long-term rescue of photoreceptors because of the short half-life of the trophic factor. Also, we have no data indicating a dose-response relationship for multiple injections of bFGF. Therefore, further "rescue" studies using bFGF should focus on efficient approaches for continuous delivery, for example, by the trans-corneal eyedrops system that we are developing.


点击右上角菜单,浏览器打开下载